Noriyuki Higo

氏名Name
Noriyuki Higo
所属・職名Affiliation, Title, etc.
Human Informatics and Interaction Research Institute, National Institute of Advanced Industrial Science and Technology (AIST), Principal Research Manager
研究室HPWebsite
一言メッセージShort Message
本プロジェクトでの研究概要Outline of the research in this project
Our previous studies in brain-damaged macaques have shown that functional and structural changes in brain areas neighboring the damage underlie the recovery of motor function. Acceleration of changes in these areas can promote motor function recovery after brain damage. In this project, we will examine the effects of neuromodulation including rTMS (repetitive transcranial magnetic stimulation) on functional and structural changes in neurons, as well as the stimulation parameters that maximize motor function recovery after brain damage. In addition, we will apply the techniques that have been validated in rodent research groups to brain-damaged macaque monkeys and evaluate their efficacy in restoring motor function. Cognitive dysfunction after brain damage is also a serious issue. This project aims to establish a macaque monkey model of cognitive dysfunction and establish technology to promote recovery not only in motor function but also in cognitive function.
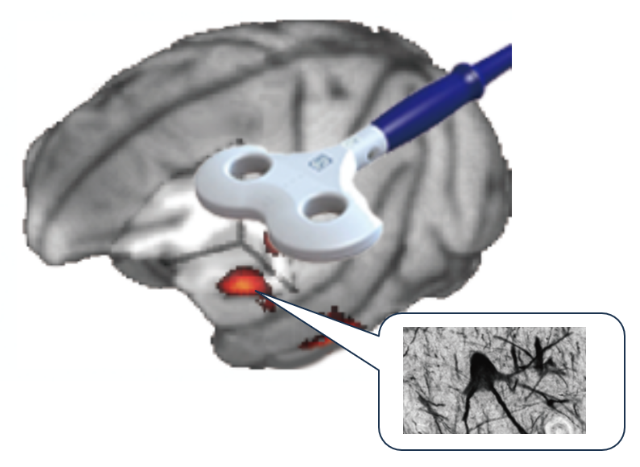
これまで主な研究内容Outline of main research so far
Damage to brain regions involved in motor output results in motor paralysis, but our previous study using a macaque monkey model have reported that subsequent rehabilitation training using grasping movements results in motor functional recovery, including dexterous movements. During functional recovery, compensatory changes in neural activity occur especially in the ventral premotor cortex. Furthermore, a new neuronal projection from this area to the subcortical motor nuclei is established. We are also developing fNIRS technology to measure brain activity changes more easily, and are aiming to develop technology to monitor functional and structural changes in the neurons during stroke rehabilitation training for confirming the effects on the brain. In addition, pain can occur after stroke, which is called as thalamic pain or central post-stroke pain and one of the sufferings for many stroke patients. Using a macaque monkey model, we have been studying the underlying functional and structural changes in the neurons and verifying the effects of neuromodulation on pain relief.
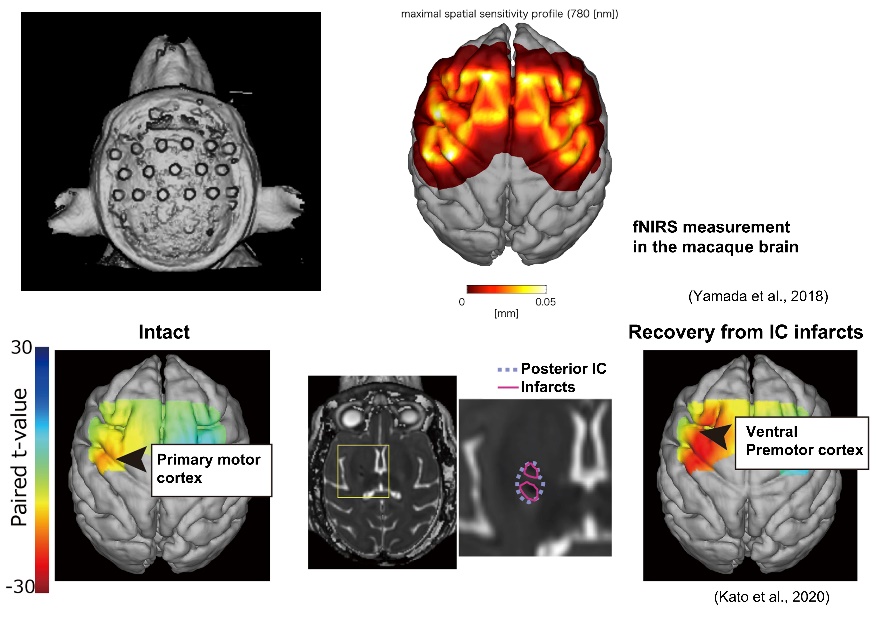
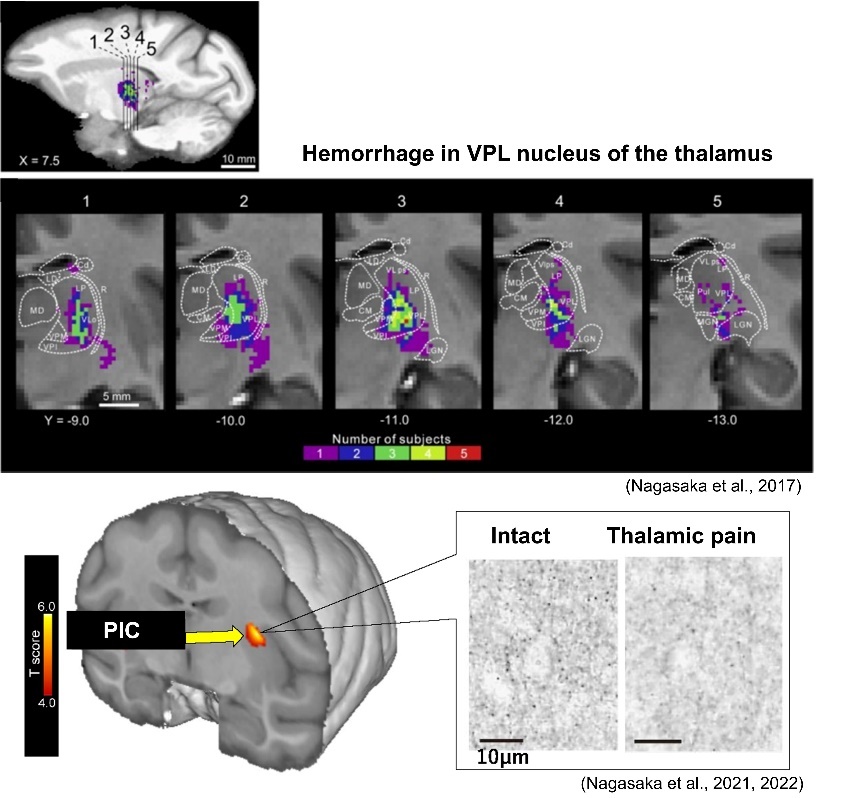
主な経歴・受賞歴等Career, Awards, etc.
Career
- 1995
- Faculty of Science, Kyoto University
- 2001
- Graduate School of Medicine, University of Tsukuba, Ph.D., Medical Science
- 2001
- Research Fellow of the Japan Society for the Promotion of Science (PD)
- 2002
- Researcher, Neuroscience Research Institute, National Institute of Advanced Industrial Science and Technology (AIST)
- 2008
- Senior Researcher, Neuroscience Research Institute, National Institute of Advanced Industrial Science and Technology (AIST)
- 2009
- JST PRESTO Research Fellow (-2013)
- 2018
- Group Leader, Human Informatics Research Institute, National Institute of Advanced Industrial Science and Technology (AIST)
- 2020
- Group Leader, Human Informatics and Interaction Research Institute, National Institute of Advanced Industrial Science and Technology (AIST)
- 2024
- Principal Research Manager, Human Informatics and Interaction Research Institute, National Institute of Advanced Industrial Science and Technology (AIST)
主要業績Major Publications
Yamamoto T, Yoshida, Y, Ose, T, Murata Y, Hayashi, T, Higo N, Cerebellar molecular signatures in non-human primates, The Journal of Comparative Neurology, 532:e25678, 2024
https://doi.org/10.1002/cne.25678
Nagasaka K, Higo N, Brain activity changes after high/low frequency stimulation in a nonhuman primate model of central post-stroke pain, Scientific Reports 14: 16527, 2024
https://doi.org/10.1038/s41598-024-67440-9
Nagasaka K, Takashima I, Matsuda K, Higo N, Pharmacological inactivation of the primate posterior insular/secondary somatosensory cortices attenuates thermal hyperalgesia, European Journal of Pain 26 (8), 1723-1731, 2022
https://doi.org/10.1002/ejp.1996
Matsuda K, Nagasaka K, Kato J, Takashima I, Higo N, Structural plasticity of motor cortices assessed by voxel-based morphometry and immunohistochemical analysis following internal capsular infarcts in macaque monkeys, Cerebral Cortex Communications 3 (4), tgac046, 2022
https://doi.org/10.1093/texcom/tgac046
Nagasaka K, Nemoto K, Takashima I, Bando D, Matsuda K, Higo N, Structural plastic changes of cortical gray matter revealed by voxel-based morphometry and histological analyses in a monkey model of central post-stroke pain, Cerebral Cortex, Volume 31, Issue 10, Pages 4439–4449, 2021
https://doi.org/10.1093/cercor/bhab098
Kato J, Y. Murata Y, Takashima I, Higo N, Time- and area-dependent macrophage/microglial responses after focal infarction of the macaque internal capsule. Neuroscience Research, Volume 170, 350-359, 2021
https://doi.org/10.1016/j.neures.2020.12.001
Kato J, Yamada T, Kawaguchi H, Matsuda K, Higo N, Functional near-infrared-spectroscopy-based measurement of changes in cortical activity in macaques during post-infarct recovery of manual dexterity. Scientific Reports, 10 (1), Article number: 6458, 1-12, 2020
https://doi.org/10.1038/s41598-020-63617-0
Nagasaka K, Takashima I, Matsuda K, Higo N, Brain activity changes in a monkey model of central post-stroke pain. Experimental Neurology, 323, 113096, 2020
https://doi.org/10.1016/j.expneurol.2019.113096
Yamamoto T, Hayashi, T, Murata Y, Ose, T, Higo N, Premotor cortical-cerebellar reorganization in a macaque model of primary motor cortical lesion and recovery. Journal of Neuroscience, 39 (43), 8484-8496, 2019
https://doi.org/10.1523/JNEUROSCI.0077-19.2019
Abe H, Jitsuki S, Nakajima W, Murata Y, Jitsuki-Takahashi A, Katsuno Y, Tada H, Sano A, Suyama K, Mochizuki N, Komori T, Masuyama H, Okuda T, Goshima Y, Higo N, Takahashi T, CRMP2-binding compound, edonerpic maleate, accelerates motor function recovery from brain damage. Science 360 (6384), 50-57, 2018
DOI: 10.1126/science.aao2300



