Yasunori Hayashi

氏名Name
Yasunori Hayashi
所属・職名Affiliation, Title, etc.
Professor, Systems Neuropharmacology
Kyoto University Graduate School of Medicine
研究室HPWebsite
一言メッセージShort Message
本プロジェクトでの研究概要Outline of the research in this project
In order to maximize the function of damaged brain regions, it is essential that reservoir neurons are appropriately integrated into neural circuits. Traditional approaches using overexpression of synaptic proteins lacked selectivity for synapses and circuits, leading to difficulties in proper information processing by the brain. To address this issue, we aim to develop a method that lowers the threshold for synaptic plasticity while preserving key characteristics such as cooperativity, input specificity, and associativity. To achieve this, we focus on the CaMK (calmodulin kinase) pathway and CREB, both of which play critical roles in promoting synaptic plasticity. Overexpression of these molecules lowers the threshold for synaptic plasticity and facilitates plastic changes. Therefore, we seek to establish a method to regulate these signaling pathways using chemogenetic and optogenetic techniques to precisely control synaptic plasticity. Subsequently, we will apply this approach to aged mice and Alzheimer’s disease model mice to evaluate its effectiveness in improving memory function. Ultimately, we aim to investigate whether similar strategies can be applied to marmosets, paving the way for future translational research.
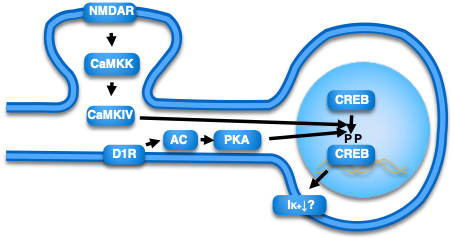
これまで主な研究内容Outline of main research so far
We have been studying long-term potentiation (LTP), a phenomenon in which transient synaptic input enhancement leads to long-term synaptic strengthening, as a cellular model of memory. To investigate this, we have utilized techniques such as gene introduction, two-photon microscopy, and FRET analysis. First, we demonstrated that AMPA receptors migrate to the synapse during LTP and found that CaMKII activation is both necessary and sufficient for this process, whereas AMPA receptor phosphorylation is not required. Additionally, we discovered that CaMKII, a kinase involved in synaptic plasticity, undergoes liquid-liquid phase separation upon activation, inducing the condensation of other proteins. We proposed that this mechanism regulates molecular translocation and accumulation at the synapse during LTP. Furthermore, we used FRET to show that LTP induces spine enlargement and promotes actin polymerization. We also found that CaMKII interacts with F-actin, and upon activation, it dissociates from F-actin, allowing actin modifications to occur during LTP.
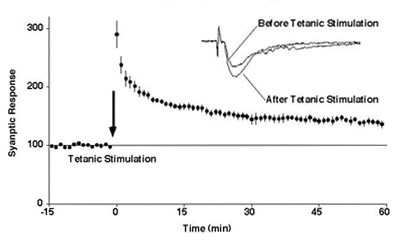
LTP in hippocampal CA1 region
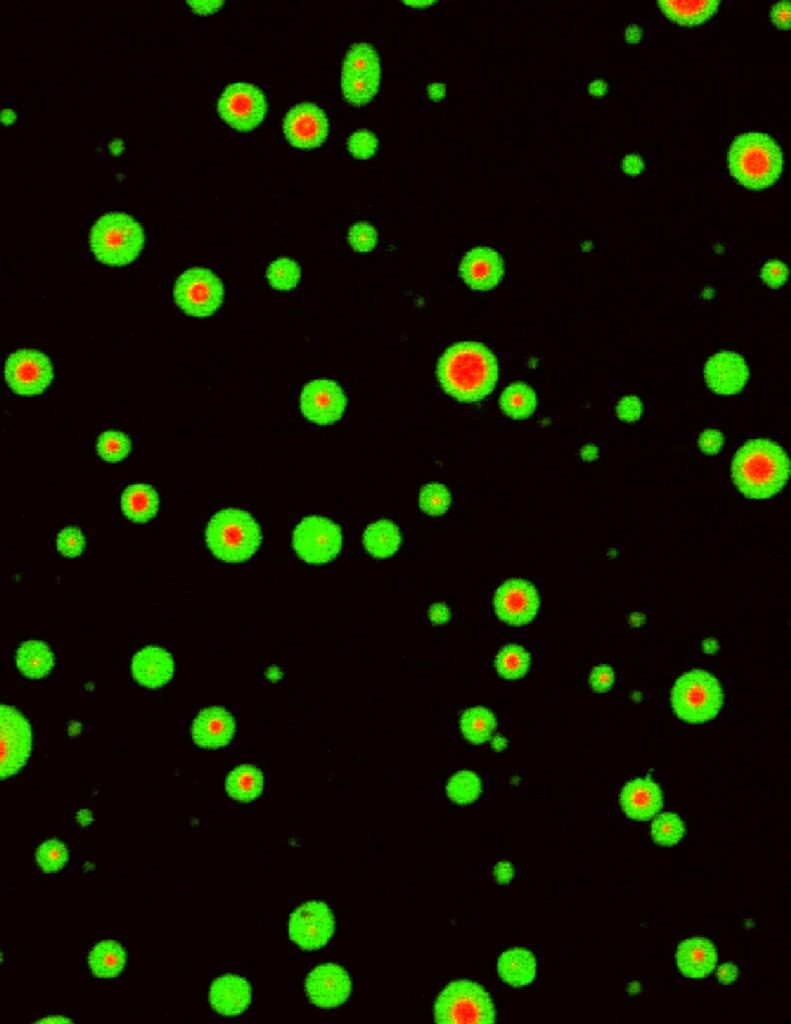
Liquid-liquid phase separation of CaMKII
Activation of CaMKII induces segregation of NMDA receptors (green) and AMPA receptor (red).
主な経歴・受賞歴等Career, Awards, etc.
Career
- 1984 - 1990
- Kyoto University Faculty of Medicine, MD
- 1990 - 1994
- Institute for Immunology and Department of Pharmacology, Kyoto University Faculty of Medicine, PhD
- 1994 - 1996
- Postdoctral Fellow, Department of Neurophysiology Institute for Brain Research, Faculty of Medicine, University of Tokyo
- 1996 - 2000
- Postdoctral Fellow, Cold Spring Harbor Laboratory
- 2000 - 2009
- Assistant Professor (joint), RIKEN-MIT Neuroscience Research Center, Massachusetts Institute of Technology
- 2009 - 2013
- Team Leader, Brain Science Institute, RIKEN
- 2013 - 2017
- Senior Team Leader, Brain Science Institute, RIKEN
- 2016 -
- Professor Kyoto University Graduate School of Medicine, Department of Pharmacology
Awards
- 2008
- JSPS Prize for Young Investigators
- 2008
- Japan Academy Medal
- 2019
- Toshihiko Tokizane Memorial Award
- 2021
- Setsuro Ebashi Award
主要業績Major Publications
Ripoli, C., Dagliyan, O., Renna, P., Pastore, F., Paciello, F., Sollazzo, R., Rinaudo, M., Battistoni, M., Martini, S., Tramutola, A., Sattin, A., Barone, E., Saneyoshi, T., Fellin, T., Hayashi, Y., and Grassi, C. (2023). Engineering memory with an extrinsically disordered kinase. Sci Adv 9, eadh1110.
https://doi.org/10.1126/sciadv.adh1110
Yasuda, R., Hayashi, Y., and Hell, J.W. (2022). CaMKII: a central molecular organizer of synaptic plasticity, learning and memory. Nat Rev Neurosci.
https://doi.org/10.1038/s41583-022-00624-2
Özden, C., Sloutsky, R., Mitsugi, T., Santos, N., Agnello, E., Gaubitz, C., Foster, J., Lapinskas, E., Esposito, E.A., Saneyoshi, T., Kelch, B.A., Garman, S.C., Hayashi, Y., and Stratton, M.M. (2022). CaMKII binds both substrates and activators at the active site. Cell reports 40, 111064.
https://doi.org/10.1016/j.celrep.2022.111064
Hosokawa, T., Liu, P.W., Cai, Q., Ferreira, J.S., Levet, F., Butler, C., Sibarita, J.B., Choquet, D., Groc, L., Hosy, E., Zhang, M., and Hayashi, Y. (2021). CaMKII activation persistently segregates postsynaptic proteins via liquid phase separation. Nat Neurosci 24, 777-785.
https://doi.org/10.1038/s41593-021-00843-3
Goto, A., Bota, A., Miya, K., Wang, J., Tsukamoto, S., Jiang, X., Hirai, D., Murayama, M., Matsuda, T., McHugh, T.J., Nagai, T., and Hayashi, Y. (2021). Stepwise synaptic plasticity events drive the early phase of memory consolidation. Science 374, 857-863.
https://doi.org/10.1126/science.abj9195
Sato, M., Mizuta, K., Islam, T., Kawano, M., Sekine, Y., Takekawa, T., Gomez-Dominguez, D., Schmidt, A., Wolf, F., Kim, K., Yamakawa, H., Ohkura, M., Lee, M.G., Fukai, T., Nakai, J., and Hayashi, Y. (2020). Distinct Mechanisms of Over-Representation of Landmarks and Rewards in the Hippocampus. Cell reports 32, 107864.
https://doi.org/10.1016/j.celrep.2020.107864
Saneyoshi, T., Matsuno, H., Suzuki, A., Murakoshi, H., Hedrick, N.G., Agnello, E., O'Connell, R., Stratton, M.M., Yasuda, R., and Hayashi, Y. (2019). Reciprocal Activation within a Kinase-Effector Complex Underlying Persistence of Structural LTP. Neuron 102, 1199-1210 e1196.
https://doi.org/10.1016/j.neuron.2019.04.012
Kim, K., Lakhanpal, G., Lu, H.E., Khan, M., Suzuki, A., Kato-Hayashi, M., Narayanan, R., Luyben, T.T., Matsuda, T., Nagai, T., Blanpied, T.A., Hayashi, Y., and Okamoto, K. (2015). A temporary gating of actin remodeling during synaptic plasticity consists of the interplay between the kinase and structural functions of CaMKII. Neuron 87, 813-826.
https://doi.org/10.1016/j.neuron.2015.07.023
Hosokawa, T., Mitsushima, D., Kaneko, R., and Hayashi, Y. (2015). Stoichiometry and phosphoisotypes of hippocampal AMPA type glutamate receptor phosphorylation. Neuron 85, 60-67.
https://doi.org/10.1016/j.neuron.2014.11.026
Bosch, M., Castro, J., Saneyoshi, T., Matsuno, H., Sur, M., and Hayashi, Y. (2014). Structural and molecular remodeling of dendritic spine substructures during long-term potentiation. Neuron 82, 444-459.
https://doi.org/10.1016/j.neuron.2014.03.021



