Study of reservoir functions that support resilience of the brain and its enhancement to overcome dementia
■Overview
The prevalence of dementia is rapidly increasing worldwide as populations age. It significantly impairs the quality of life for patients and their families and imposes a heavy burden on healthcare and social systems.
Conventional treatments for dementia, especially Alzheimer's disease, have primarily focused on eliminating abnormally accumulated proteins such as amyloid-beta and tau. However, it has become increasingly clear that these treatments alone are insufficient to prevent the onset or progression of dementia.
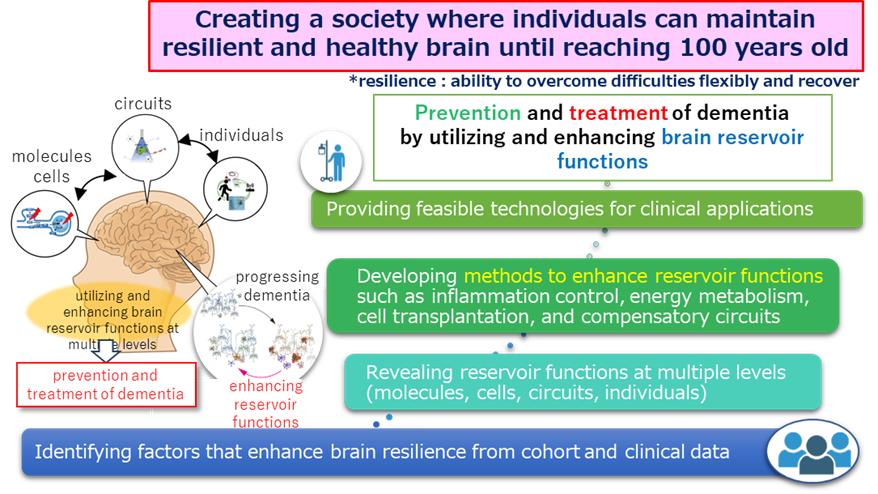
This project takes a completely new approach to tackling dementia. Instead of targeting pathological changes directly, we focus on the brain’s “reservoir function”—its compensatory mechanisms and alternative neural pathways that help preserve cognitive function even in the face of pathological changes. We aim to clarify the mechanisms underlying this reservoir function and to develop practical strategies for enhancing it.
By strengthening the reservoir function, we seek to minimize cognitive decline even when pathological changes occur, prevent the onset of dementia during the prodromal stage, and improve cognitive function even after dementia has developed. Ultimately, we aim to help build a society where individuals can maintain a resilient, dementia-resistant brain well into old age—up to 100 years and beyond.
■Research framework
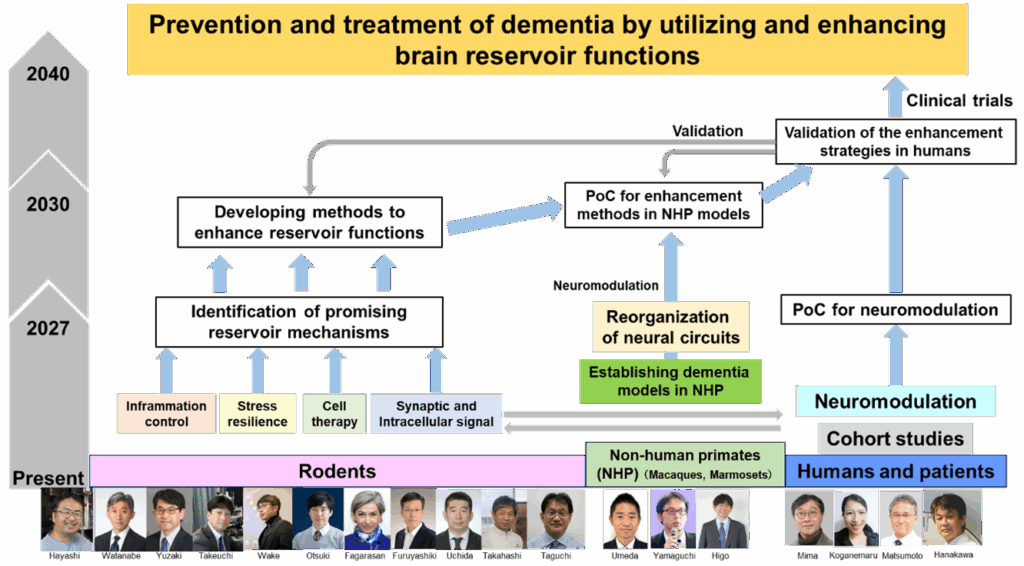
The reservoir function is supported by various biological foundations, including neuronal and synaptic plasticity, regulation of the immune system and inflammation, stress resistance, and energy metabolism. Also, potential methods for enhancing the reservoir function include drug discovery, cell therapy, and brain stimulation (neuromodulation). This project brings together leading researchers from each of these fields to collaborate closely and advance the research. Furthermore, by using human subjects, rodents, and non-human primates, we will employ bidirectional and translational research approaches: clarifying mechanisms discovered in humans using rodents, and validating enhancement methods developed in rodents using primates and humans.
■Goals for 2040
1. Finding promising reservoir functions to treat dementia.
2. Developing methods to enhance reservoir functions. (e.g., through drug discovery, cell therapy, or brain stimulation).
3. Creating prevention and treatment of dementia by utilizing and enhancing brain reservoir functions.
